Sinoright Blog
Malonodinitrile
MALONONITRILE
(dinitrile of malonic acid, malonitrile, propandinitrile, dicyanomethane) CH2(CN)2, mol. m. 66.07; colorless. crystals; t. pl. 31,6-32,4 °C, t. kip. 218-219°C;d435 1,0494; nD35 1,4130; m 11,9.10-30 Kl. m; C°p110 kJ/mol; DH0cgor -16.55 kJ/mol, DH0 pl 10.06 kJ/mol, DH0 obr -265.9 kJ/mol; S0298131 J/(mol. K); steam pressure at 60 ° C 200 Pa; h2.85 MPa X s (32.7 ° C); g 47.9 mN/ m (37.5 ° C); solubility (g in 100 g): in water 13.3, ethanol 40, diethyl ether 20. M. has a high reactivity. ability and is widely used in organ synthesis. Condensation of it with carbonyl compounds. under the conditions of the Knevenagel reaction - a general method for obtaining dicyanoolefins:CH2(CN)2 + RR'C = O : RR'C = C(CN)2
This reaction is used in the synthesis of 7,7,8,8-tetracyanoquinodimethane and some dyes. Soed. with dicyanomethylene groups, they are also formed at the reactions of M. with derivatives of quinolinium, tropilia and cyclopropenylium, for example.:
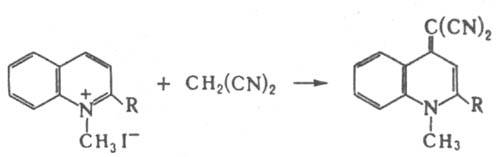
Bromination of M. leads to dibromalononitrile Bg2 With (CN)2 - gap. the product in the synthesis of tetracyanoethylene, the reaction with chlorocyanum - to the cyanoform HC(CN)3. Under the action of the bases, M. dimerizes into 2-amino-1,1,3-tricyanopropene, the reactions of which lead to different heterocyclic derivatives. connections. In the M. industry, they synthesize the interaction. chlorocyanin with an excess of acetonitrile:
ClCN + CH3CN : CH2(CN)2 +НСl
He M. B. is also obtained by dehydration of cyanoacetamide by the action of P2O5 or ROSl3. Qualities. The analysis of M. is based on the formation of colored compounds. in combination with phenanthrenquinone or nitroprusside Na. Quantities. the determination is carried out using a GC or a non-aqueous potentiometer. titration. M. is used in the synthesis of lek. drugs, herbicides, fungicides, defoliants, chlorobenzalmalonodinitrile, dyes and pigments, etc. It is also used as a crosslinking agent for polymers, an additive to lubricating oils that stabilizes their viscosity, and a complexing agent in the extraction of gold. M. is highly toxic; LD 50 20-60 mg/kg (rats, mice, orally). Vol. 112°C. Lit.: Freeman F., "Chem. Rev.", 1969, v. 69, No. 5, p. 591-624; Fatiadi A. J, "Synthesis", 1978, No. 3, p. 165-204; 1978, No. 4, p. 241-82. D. V. Ioffe.
1)Name: Malonodinitrile (Synonyms: Malonodinitrile, Malonic acid Dinitrile, Malononitrile, Malonodinitrile, cyanoacetonitrile, dicyanomethane, malonic acid dinitrile, malononitrile, methylene cyanide, propanedinitrile)
2)CAS Number: 109-77-3
3)Molecular Weight: 66.06
4)Molecular Formula: C3H2N2
5)Specification:
Batch number 20150724
Quality indicators Normative indicators Final indicators
Appearance Colorless or yellowish crystalline powder Colorless crystalline powder
Melting point (°C) 31-32 31.4
The content of the main substance (%) ≥99 99.56
Acid number (%) ≤0.3 0.1
Combustion residue (%) ≤0.05 0.03
6)Packaging: 200 kg metal barrels
Malononitrile, also propandinitrile or malonodinitrile, is a nitrile with the formula CH2(CN) 2.
Malononitrile is relatively acidic, with a pKa of 11 in water. This allows it to be used in the condensation of Knevenagel, for example, in the production of CS gas :
In related chemistry, malononitrile is a suitable starting material for the Gewald reaction, where nitrile condenses with ketone or aldehyde in the presence of elemental sulfur and base to form 2-amino thiophene.
